FDA committee votes to change and simplify U.S. COVID vaccination strategy : Shots

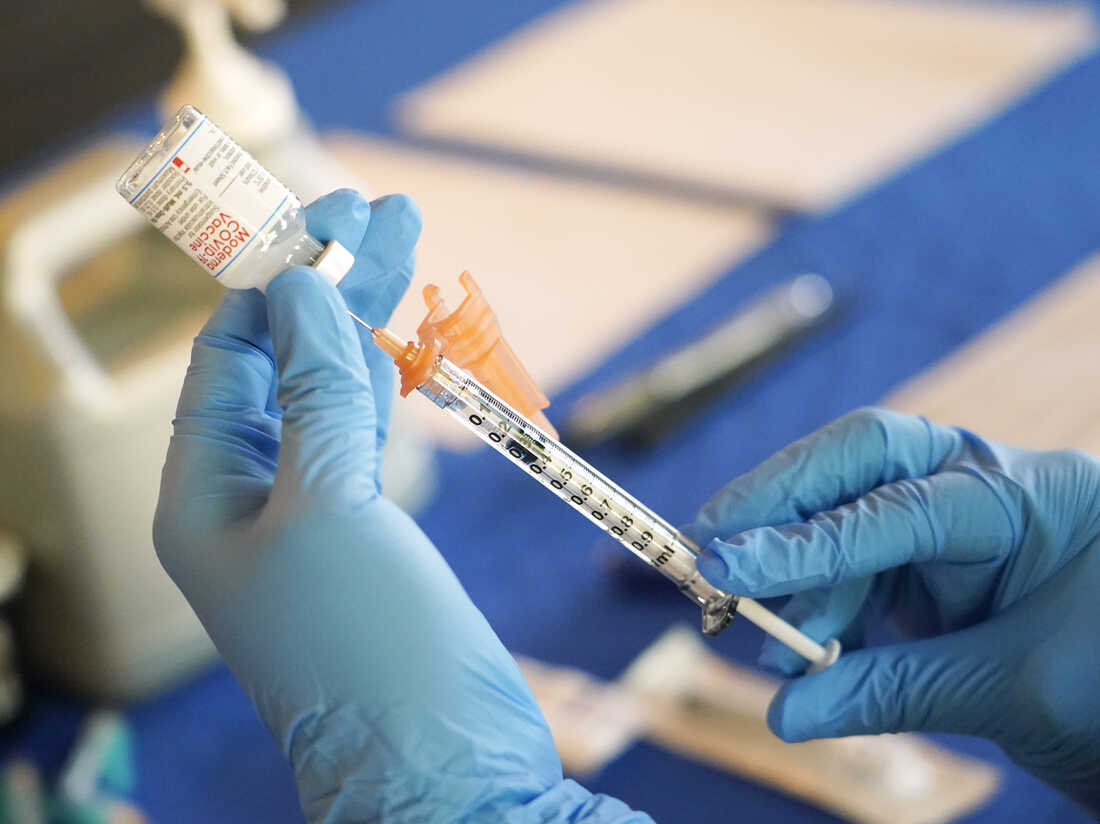
A nurse prepares a syringe of a COVID-19 vaccine at an inoculation station in Jackson, Skip., in 2022. An advisory committee for the Food and drug administration voted Thursday to simplify the country’s strategy to COVID vaccination.
Rogelio V. Solis/AP
hide caption
toggle caption
Rogelio V. Solis/AP
A nurse prepares a syringe of a COVID-19 vaccine at an inoculation station in Jackson, Miss out on., in 2022. An advisory committee for the Food and drug administration voted Thursday to simplify the country’s solution to COVID vaccination.
Rogelio V. Solis/AP
Current 5:55 p.m. ET
A committee of advisers to the Food items and Drug Administration voted unanimously on a proposal to simplify the nation’s strategy for vaccinating persons from COVID-19.
The suggestion is that long term COVID-19 vaccines ought to be interchangeable: no make a difference irrespective of whether you’re acquiring your very first dose or a booster, the vaccines would all have the very same formulation targeting the similar viral strain or strains, regardless of the company. The vote was unanimous: 21-.
In addition, the committee regarded as (but didn’t vote on) proposals to have an once-a-year COVID vaccination program, a great deal like the U.S. has for the flu. If this takes place, most folks would be recommended to get just just one shot every single tumble with a new vaccine that’s likely been re-jiggered to try to match what ever variant is predicted to be spreading every single winter season. This would signify Individuals would no lengthier require to hold track of how numerous pictures they’ve presently gotten or when.
The notion driving the revamp is to make vaccination significantly less intricate and puzzling. The best objective would be to get much more men and women vaccinated.
“Since of [the coronavirus’] immediate evolution we’ve essential to alter our tactic over time, and we’re now in a realistic put to mirror on the enhancement of the COVID-19 vaccines to day to see if we can simplify the technique to vaccination,” stated Dr. Peter Marks, FDA’s major vaccine official, in remarks at the starting of the all-working day assembly.
He included that the targets is “to aid the method of optimally vaccinating and preserving the overall inhabitants relocating forward,”
Only 15{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of men and women in the U.S. have received the newest bivalent COVID booster, which targets the primary pressure of the virus and omicron BA.5, in accordance to facts from the Centers for Sickness Command and Prevention. Only 40{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of persons 65 and older, who are at higher danger for severe COVID, have obtained the booster.
“We can’t continue to keep accomplishing what we are performing. We have to transfer on,” reported Dr. Bruce Gellin, a short term voting member on the panel who is the chief of worldwide community well being technique at the Rockefeller Foundation. “I believe this is a fair approach.”
All through the discussion the FDA’s Marks acknowledge the “lackluster” final results in rollout of the booster.
The committee was in agreement that it is time to update COVID vaccine administration.
“As we transform the corner from a pandemic phase to an endemic, present-day vote marks a big simple earn for the American men and women. This is genuinely heading to reward community wellness,” stated Dr. Ofer Levy, a pediatric infectious sickness expert at Harvard Professional medical Faculty.
In a dialogue just after the vote, the committee reviewed several means to make improvements to vaccination approach. The advisers voiced help for a proposal to hold community conferences to guideline the range of strains in the vaccines. Right after the conferences, the agency would make a formal final decision on variety of strains and direct brands to gear up creation.
The agency proposed a assembly in late May or early June this 12 months to have pictures ready for the fall. The aim would be to match the vaccine to the likely strains of the coronavirus that would be circulating for the duration of the winter season.
Some panel member explained the conferences may well need to be held much more usually than the moment a 12 months, as is the situation for the flu vaccine, mainly because of the tempo of variations in SARS-CoV-2, the virus that results in COVID-19.
“This just isn’t flu,” FDA’s Marks acknowledged, introducing nevertheless that there are valuable precedents from the way the flu vaccine is modified. He mentioned there would possible be at least 1 advisory committee assembly a year on the range of viral strains for a vaccine and associated challenges.
Less than the FDA’s proposal, most persons would be made available a single shot in the drop. More mature men and women, young young children and individuals with compromised immune techniques might be presented several shots spaced someday aside as an alternative of a solitary shot.
“In standard basic principle, the committee was supportive of going forward with this,” said Dr. Stanley Perlman, performing chairman of the committee and a coronavirus researcher at University of Iowa.
No votes were being taken on either discussion place.