Study reveals inaccurate label claims on unregulated CBD products

Recent scientific studies have highlighted the therapeutic benefits of cannabidiol (CBD), which is a non-intoxicating element of Cannabis sativa. Previously, the affiliation of Δ9-tetrahydrocannabinol (Δ9-THC) with CBD limited studies on its medicinal positive aspects.
The Agricultural Improvement Act of 2018 (2018 Farm Bill) legalized the use of goods from the plant pieces of Hashish sativa, which is also acknowledged as hemp. This led to the fast advancement of the CBD field.
Review: Label accuracy of unregulated cannabidiol (CBD) items: calculated concentration vs. label declare. Picture Credit rating: IRA_EVVA / Shutterstock.com
Background
In June 2018, a purified oral remedy of CBD regarded as Epidolex® been given approval by the United States Food items and Drug Administration (Fda) for the therapy of three kinds of epilepsy.
However, except for Epidolex®, other CBD solutions keep on being unregulated in the U.S. In June 2019, the Food and drug administration held a typical general public listening to to hear worries from researchers concerning CBD regulation.
A lot of research groups, alongside with the Fda, have identified various challenges with label precision for numerous CBD goods. This concern was not restricted to the U.S., as equivalent reports have also been posted in the Netherlands, United Kingdom, and Italy.
A new Journal of Cannabis Investigate study aimed to establish the CBD information in various products and review it to their label promises. The products and solutions assessed in this review have been obtained from quite a few merchants in Central Kentucky, as properly as online merchants, amongst April 2, 2021, and May 9, 2021.
About the examine
The recent review involved a whole of 80 samples, 44 of which have been ordered from online vendors that were U.S.-based. The remaining 36 samples had been purchased from area retailers within just Central Kentucky.
Taken jointly, the samples integrated in this analyze have been created by 51 diverse makes. Epidiolex® was also employed as a favourable command. All CBD merchandise have been tested right away upon opening and right before their expiration dates.
To determine label accuracy, a ±10{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of the allowable variance was employed. The products and solutions with CBD focus exceeding 110{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of the labeled price ended up viewed as underneath-labeled, though these with a lot less than 90{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of their labeled CBD material ended up deemed about-labeled. Items with a CBD concentration that was involving 90-110{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of their labeled CBD information were being viewed as to be precisely labeled.
Thereafter, all samples had been geared up and analyzed making use of liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Study conclusions
Out of the 80 analyzed samples, 31{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} were being beneath-labeled, 54{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} were being labeled accurately, and 15{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} had been about-labeled. Out of the 44 merchandise that were being bought on-line, 25{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} had been under-labeled, 61{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} have been labeled precisely, and 14{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} ended up more than-labeled. For the 36 products that had been ordered locally, 39{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} were being less than-labeled, 17{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} were being in excess of-labeled, and 44{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} have been labeled precisely.
The CBD concentrations ranged from 2.9 to 61.3 mg/mL for all unregulated samples, with labeled CBD concentrations ranging from 17-159{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d}. The average amount of money of CBD for beneath-labeled items was 121{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} of the label assert, as in contrast to 61{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} in in excess of-labeled merchandise.
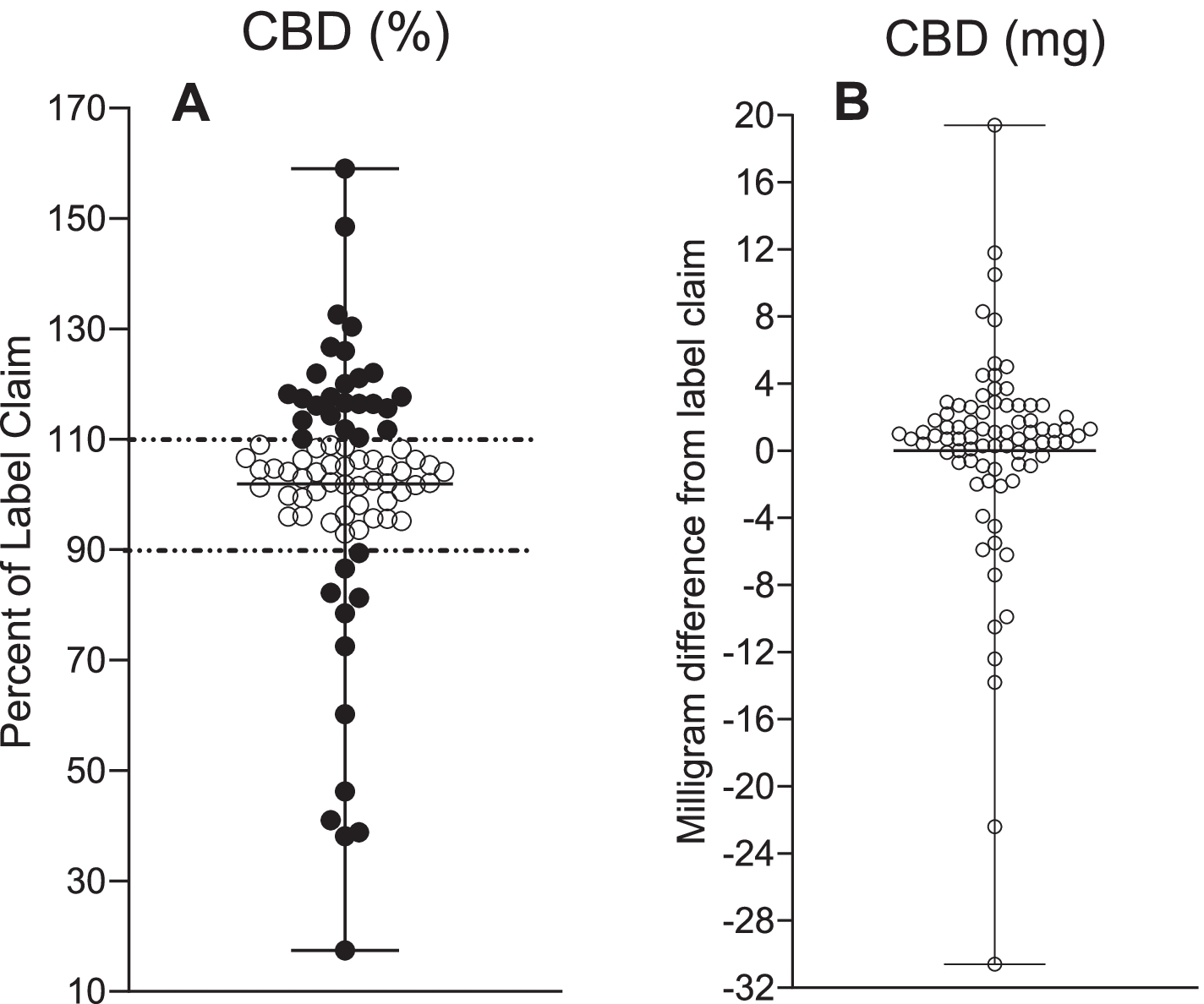 CBD measurements in 80 commercially available CBD oil goods and Epidiolex®. A The proportion of CBD label declare content material with ± 10{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} tolerance denoting under-labeling (> 110{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d}) and in excess of-labeling (< 90{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d}). B Deviation from CBD label claim in milligram
CBD measurements in 80 commercially available CBD oil goods and Epidiolex®. A The proportion of CBD label declare content material with ± 10{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d} tolerance denoting under-labeling (> 110{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d}) and in excess of-labeling (< 90{b574a629d83ad7698d9c0ca2d3a10ad895e8e51aa97c347fc42e9508f0e4325d}). B Deviation from CBD label claim in milligram
Conclusions
The current study provides evidence that CBD products from several countries around the world often had inconsistent CBD concentrations with their label claims. Such inaccurate labelings can pose safety risks to consumers who use CBD products for medical treatments.
Thus, clear regulations from both the federal and state agencies, as well as good manufacturing practices, are required to guarantee that the label claims of CBD products are accurate.
Limitations
The study has certain limitations. First, the study included only hemp-derived oil products. Second, only CBD concentrations were reported, while other cannabinoid concentrations were not reported. The current study also did not involve a formal sampling protocol.
Journal reference:
- Johnson, E., Kilgore, M., & Babalonis, S. (2022). Label accuracy of unregulated cannabidiol (CBD) products: measured concentration vs. label claim. Journal of Cannabis Research. doi:10.1186/s42238-022-00140-1.